Heat
Heat energy is defined as the sum of total kinetic energy of all the molecules of the substance. Its SI unit is Joule (J).
Heat
Heat energy is defined as the sum of total kinetic energy of all the molecules of the substance. Its SI unit is Joule (J).
Define 1 joule heat energy
One joule heat energy is defined as the amount of the heat energy required to raise the temperature of any substance of mass 1 kg through 1°C having specific heat capacity 1 J/kg°C.
Define 1 calorie heat
The amount of heat energy required to raise the temperature of 1 gm pure water through one degree Celsius temperature is called one calorie heat.
Relation between Joule and Calorie
1 Calorie = 4.2 Joule
Factors on which heat energy depends
The factors are:-
- Mass or total no. of molecules of the object.
- Kinetic energy of each molecules of the object.
Relation between heat content, K.E. and mass
Relation:
Heat energy ∝ average K.E. of the molecule × mass
Define temperature
Temperature of any object is defined as the average kinetic energy of all the molecules of the substance. Its SI unit is Kelvin (K).
Factor on which temperature depends
Average kinetic energy of each molecules of the object.
Devices to measure heat and temperature
The device which is used to measure heat energy is calorimeter and temperature is thermometer.
CGS unit and conversion
Calorie is the CGS unit of heat energy. According to the relation,
4.2 Joule = 1 calorie
or, 1J = 1/4.2 Calorie
or, 100 J = 1/4.2 × 100 Calorie
= 23.81 Calorie
Relation among heat, mass and K.E.
The quantity of heat contained by an object is directly proportional to the average kinetic energy of molecules and mass of the object.
Quantity of heat ∝ average K.E. of molecules × Mass of the object
Direction of heat flow
Heat flows from the object of higher temperature to the lower temperature.
Relationship between molecular vibration and temperature
When a molecule is heated, the vibration of the molecule increases and then the K.E. of the molecule increases. Since K.E. is directly proportional to the temperature, temperature also increases.
Temperature ∝ vibration of the molecules
Differentiate between Heat and Temperature
What is Thermometer?
Thermometer is a device which is used to measure the temperature.
Principle of Thermometer
The principle of thermometer is every liquid expands on heating and contracts on cooling.
Fixed Points of Thermometer
The lower fixed point is the temperature of pure melting ice at sea level whereas the upper fixed point is the temperature of boiling water at sea level. The lower fixed point in Celsius is 0°C whereas upper fixed point is 100°C.
Temperature Scales
Different scales used to measure temperature are Celsius scale, Fahrenheit scale, Kelvin scale and Rankin Scale.
(C - 0)/100 = (F - 32)/180 = (K - 273)/100 = (R - 0)/100
When are °C and °F equal?
Let x °C = x °F
We have, (C-0)/100 = (F-32)/180
or, (x-0)/100 = (x-32)/180
Or, 18x = 10x – 320
Or, 8x = -320
Or, x = -40
Hence, -40°C = -40°F
Thermometric Liquid
The liquid kept inside the thermometer is called thermometric liquid. The commonly used thermometric liquids are mercury and alcohol.
Why Mercury is used as Thermometric Liquid
Mercury is used as a thermometric liquid because of its following properties:-
- It is opaque, bright and silvery in color.
- It can expand and contract due to small change in temperature.
- It does not wet the walls of the capillary tube.
Clinical Thermometer
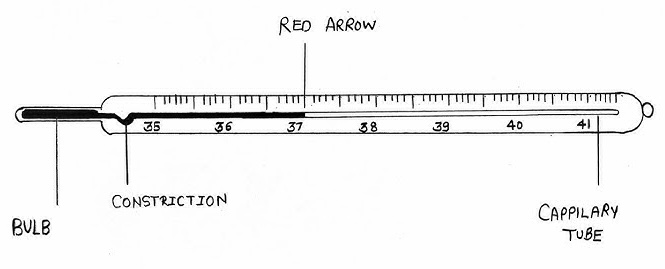
A clinical thermometer is a specialized medical device designed to accurately measure the human body temperature. It is a crucial tool for diagnosing fevers and monitoring health conditions.
Working Mechanism
It operates on the principle of thermal expansion. Traditionally, it contained a narrow, sealed glass tube with a bulb at one end filled with mercury. When placed in the mouth, armpit, or rectum, the heat from the body causes the mercury to expand, forcing it to rise up the narrow constriction (kink) in the tube. This kink prevents the mercury from falling back immediately after removal, allowing for a reading to be taken.
Scale
The scale is marked from 35°C (95°F) to 42°C (107.6°F). This limited range covers all possible variations in human body temperature, which normally hovers around 37°C (98.6°F). The scale is finely divided into 0.1°C or 0.2°F divisions for high precision.
Applications
- Diagnosing Fever: The primary use is to check for an elevated body temperature, a key symptom of infection and illness.
- Monitoring Health: Tracking the progress of a fever (e.g., to see if medication is working).
- Preventive Healthcare: Routine check-ups to ensure normal body function.
- Hospitals and Homes: Used extensively by medical professionals and for personal home care.
Laboratory Thermometer
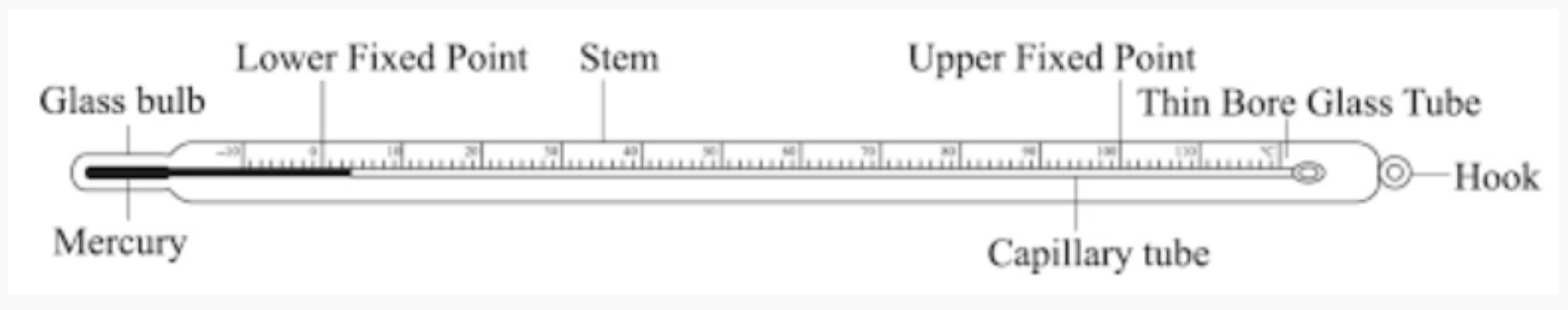
A laboratory thermometer is a device designed for scientific experiments to measure temperature with a high degree of precision. It is used for a wide range of substances, not just the human body.
Working Mechanism
Traditional laboratory thermometers operate on the principle of thermal expansion, similar to clinical thermometers. A liquid (historically mercury or now often a safe organic red liquid) is sealed in a glass tube with a bulb at the end. When heated, the liquid expands and rises up the narrow capillary tube to indicate the temperature. Unlike clinical thermometers, they do not have a constriction (kink), so the level falls once removed from the heat source. Modern versions may be digital, using a thermistor probe for electronic measurement.
Scale
The scale is a key differentiating feature. It has a much wider range than a clinical thermometer, typically from -10°C to 110°C or even broader (e.g., -200°C to over 500°C for specialized types). This allows it to measure the boiling and freezing points of various substances. The graduations are precise, often marked in whole degrees or 0.1° or 0.2° divisions for accuracy.
Applications
- Scientific Experiments: Measuring the temperature of chemicals, solvents, and mixtures during reactions in chemistry and biology labs.
- Educational Use: Demonstrating principles of heat and thermodynamics in school and university science classes.
- Industrial Use: Monitoring temperatures in manufacturing and quality control processes.
- Meteorology: Used in weather stations to measure ambient air temperature (often in a Stevenson screen).
Digital Thermometer
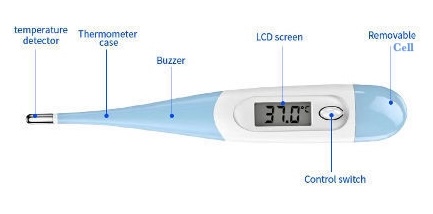
A digital thermometer is a modern electronic device used to measure temperature. It has largely replaced traditional mercury thermometers due to its safety, speed, and ease of reading.
Working Mechanism
It operates using a thermistor, a type of electrical resistor whose resistance changes predictably with temperature. The thermometer's probe is placed at the measurement site. The internal microprocessor detects the change in resistance, converts it into a temperature value, and displays it digitally on a liquid crystal display (LCD) screen. Most models signal the end of the measurement with an audible beep.
Scale
Digital thermometers are highly versatile and can be programmed to display temperature in Celsius (°C) or Fahrenheit (°F). Their electronic range is typically broader than a clinical thermometer, often from about 32°C (90°F) to 43°C (110°F) or wider, making them suitable for various applications beyond just human body temperature.
Applications
- Medical Use: Measuring human body temperature orally, rectally, or axillarily (underarm). Some designs are specifically for tympanic (ear) or temporal (forehead) use.
- Domestic Use: A common household item for checking for fever in adults and children. Their unbreakable design and lack of glass or mercury make them safe for home use.
- Culinary Use: Certain models are used to measure the temperature of food, liquids, or cooking surfaces.
- Aquarium and Laboratory Use: Their quick response and digital accuracy make them useful for monitoring environmental temperatures.
Maximum and Minimum Thermometer
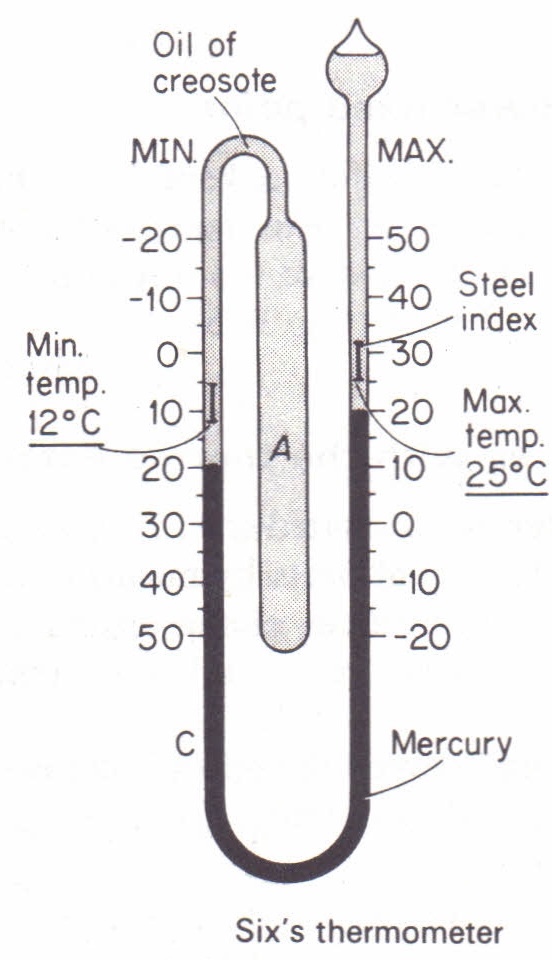
The Maximum and Minimum thermometer is a specialized instrument designed to record the highest and lowest temperatures reached over a specific period, usually 24 hours. It is invaluable in meteorology, horticulture, and agriculture for tracking temperature fluctuations.
Working Principle
It operates on the principle of thermal expansion of a liquid and the physical displacement of indicators. Unlike a standard thermometer that shows the current temperature, this thermometer has two separate indicators that are pushed by the expanding fluid and remain at their extreme positions until manually reset.
Applications
- Meteorology and Weather Stations: Its primary use is in weather forecasting and climate recording. It provides crucial data on daily and nightly temperature ranges.
- Agriculture and Horticulture: Farmers and gardeners use it to monitor conditions critical for plant growth, to predict frosts, and to protect sensitive crops.
- Greenhouse Management: Essential for maintaining optimal and stable temperature ranges for plants inside a greenhouse.
- Educational Purposes: Used in schools to teach students about weather patterns, temperature extremes, and the properties of thermal expansion.
Advantages and Disadvantages
Advantages
- It provides both maximum and minimum data simultaneously.
- It has simple mechanical design with no need for power.
- It is highly accurate for its intended purpose.
Disadvantages
- It requires manual resetting every day.
- It has fragile glass construction; must be mounted horizontally.
- Reading the markers can sometimes be prone to parallax error.
Infrared Thermometer
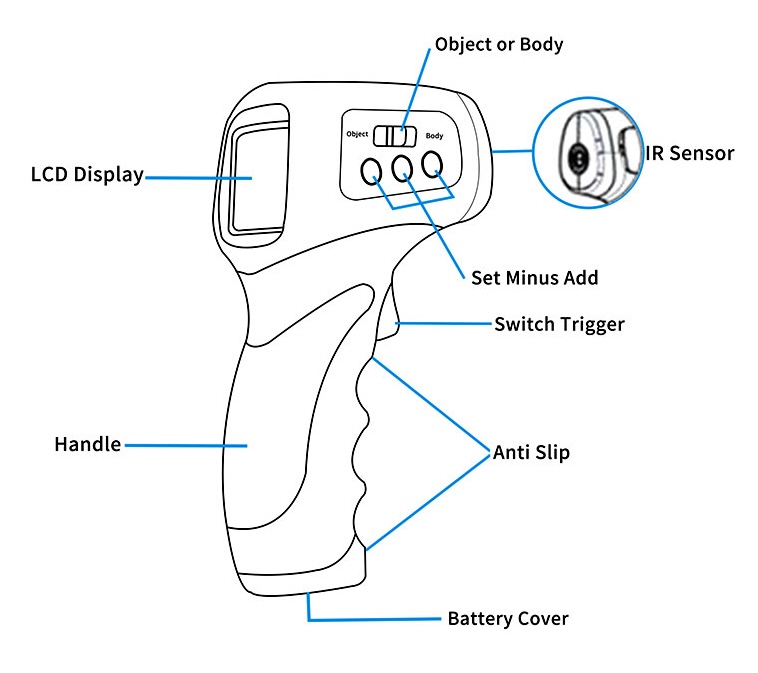
An Infrared (IR) thermometer, also known as a pyrometer or non-contact thermometer, is a device that measures the temperature of a surface from a distance by detecting the infrared radiation it emits. It is renowned for its speed, safety, and ability to measure objects that are difficult to access, moving, or extremely hot.
Working Principle
It operates on the fundamental principle of black body radiation. All objects with a temperature above absolute zero (-273.15°C) emit infrared radiation. The intensity and wavelength of this radiation are directly proportional to the object's temperature. The IR thermometer captures this radiation and converts it into an electrical signal, which is then translated into a temperature reading.
Applications
- Medical and Healthcare: Rapid, non-contact screening of body temperature (e.g., temporal artery or tympanic thermometers), minimizing cross-contamination.
- Industrial Maintenance: Identifying overheating electrical components (circuit breakers, connections), mechanical parts (bearings, motors), and steam traps to predict failures.
- Food Safety: Checking the surface temperature of food, grills, refrigerators, and storage areas without making contact, which is crucial for HACCP compliance.
- HVAC: Diagnosing issues with heating and air conditioning systems by measuring vent temperatures, coil performance, and insulation efficiency.
- Automotive: Checking tyre tread temperature, brake disc temperature, and engine performance.
- Scientific Research: Measuring temperatures in experiments where contact is impossible or would interfere with the process.
Advantages and Disadvantages
Advantages
- Non-contact: Measures from a distance; safe for hazardous, moving, or electrically live objects.
- Very Fast: Provides readings in seconds or less.
- Hygiene: Eliminates risk of cross-contamination, ideal for medical and food industry use.
- Durable: No moving parts that can wear out from repeated use.
Disadvantages
- Surface Measurement Only: Measures the outer surface temperature, not the internal temperature.
- Emissivity Errors: Accuracy is highly dependent on correct emissivity settings.
- Obstructions Affected: Affected by dust, steam, smoke, and glass, which can block or alter IR radiation.
- Cost: High-quality, accurate models can be more expensive than simple contact thermometers.
Why Alcohol is used as Thermometric Liquid
Alcohol is used as a thermometric liquid because of its following properties:-
- It can expand and contract due to small change in temperature.
- Its expansion rate is about 7 times than that of mercury.
- Its freezing point -117°C, so very useful to measure low temperature.
Thermometers for Different Temperatures
Mercury thermometer is used to measure high temperature because it can measure temperature from -39°C to 357°C, whereas alcohol thermometer is used to measure low temperature because it can measure temperature from -117°C to + 78°C.
Thermometer Ranges
Mercury Thermometer
–39°C to 357°C
Alcohol Thermometer
–117°C to 78°C
Best Thermometer for Mountain Climbing
While climbing the mountains, alcohol thermometer is best because it can measure the low temperature up to –117°C.
Limitation of Alcohol Thermometer
The boiling point of the alcohol is 78°C whereas that of water is 100°C. Hence alcohol boils faster than water when it is used to measure the temperature. So that it can't be used to measure temperature of the boiling water.
Effects of Heat
Different effects of the heat energy are:-
- Change in temperature.
- Change in volume.
- Change in state.
- Change in solubility.
- Chemical change or chemical reaction.
Anomalous Expansion of Water
All liquids expand on heating and contracts on cooling but when water is heated from 0°C to 4°C its volume decreases instead of increasing and density increases so, water has maximum density and minimum volume at 4°C. This is called anomalous expansion of water. But above 4°C, it expands normally. This can be explained by the following graph.
[Graph showing anomalous expansion of water would be placed here]
Consequences of Anomalous Expansion of Water
Different consequences of anomalous expansion of water are as follows:-
- Aquatic animals can survive in a pond in a cold region although its outer surface is frozen.
- Water pipe gets burst in winter season in a cold reason.
- Water starts to freeze from the top whereas ghee starts to freeze from the bottom.
Water at 4°C - Heating and Cooling
When a glass filled with water completely at 4°C is heated or cooled it overflows. This is because due to anomalous expansion of water, water has high density and least volume at 4°C, when it is heated it's volume increases due to normal expansion of water whereas when it is cooled it's volume increases due to anomalous expansion of water.
Water Heated from 0°C to 10°C
When water is heated from 0°C to 4°C its volume decreases and density increases according to the anomalous expansion of water but above 4°C, water shows normal expansion. i.e. volume increases and density decreases. This can be shown by the following graph:
[Graph showing volume and density changes of water from 0°C to 10°C would be placed here]
Advantage for Aquatic Animals
The advantage of anomalous expansion of water for the aquatic animals is, they can live freely in a pond when outer surface is frozen. When temperature falls to 0°C in cold region during winter, the upper surface of water changes into ice. Since the density of ice is less than that of water, it floats. But due to the anomalous expansion of water, the water molecules having temperature 1°C to 4°C lie in layers below ice. The ice surface acts as a bad conductor of heat so that heat cannot escape out of the water body. Thus, water is prevented from freezing and aquatic animals can survive in it.
[Image showing aquatic animals under ice would be placed here]
Give Reasons
a) Water pipe gets burst in winter season in a cold region.
In the cold region in winter season, the surrounding temperature falls to 0°C. When the surrounding temperature falls to 0°C, water inside the pipe changes to ice. The ice occupies more space but the pipe is contracted due to excessive cold. Thus a high pressure is created and pipe gets burst.
b) The density of water is measured at 4°C.
Due to anomalous expansion of water, water has maximum density at 4°C, which is equal to 1g/cm³. Hence density is measured at 4°C.
c) Liquid food in very cold region is prevented from freezing if it is placed inside water.
Due to anomalous expansion of water in cold region water at 4°C being heavier lies below the surface of the water hence this temperature prevents the liquid food from freezing.
d) Water starts to freeze from the top whereas ghee/wax starts to freeze from the bottom.
Due to anomalous expansion of water when water is placed in a vessel, different layers of water is formed with 0°C, 1°C, 2°C, 3°C, 4°C from top to bottom. As 4°C has higher density so, water starts to freeze from top but for other liquids like ghee or wax they shows normal behavior. So, different layers with higher temperature and bottom contains 0°C liquid. So, liquid starts to freeze from bottom.
e) Vegetables and fruits get damage during too much cold.
During too much cold condition, temperature falls below 4°C. So, water pressure inside fruits and vegetables expands may burst and other components may be damaged due to expansion of water inside them.
f) Soft drink bottles (like coke) are not completely filled.
Soft drink bottles must be kept in freezing chamber of refrigerator for cooling purpose. During this the water present in Soft drink expands on cooling from 4°C to 0°C and occupies the empty space. This prevent the bottles from bursting. Therefore, soft drink bottles are not completely filled to prevent them from bursting while cooling in refrigerator.
g) We need warm clothes in winter.
In winter season, the atmospheric temperature falls below our normal body temperature (37°C). Due to this difference in temperature heat energy starts to flow from body to the surrounding. So, our body cools. So, to prevent our body from excessive cooling, we need warm clothes in winter.
h) Metal antimony behaves like water.
Similar to the anomalous expansion of water metal antimony also expands on cooling and contracts at first when heated above the melting point.
i) We sweat in summer.
In summer season, atmospheric temperature goes up higher than our normal body temperature (37°C) so, heat energy starts to flow from surroundings to our body. So, to maintain constant temperature body starts to lose temperature in the form of sweat.
j) Solid melts on heating.
The kinetic energy of the vibrating solid increases when a solid is heated because the molecule gets extra heat energy for vibration. Due to this, the intermolecular force of attraction between the molecules decreases. When the solid is further heated, the solid starts melting due to vey weak intermolecular attraction.
k) A new quilt is felt warmer than old one.
A new quilt is thicker than the old one. The thicker quilt contains more spaces which get filled with more air as compared to that of the old quilt. Since air is bad conductor of heat, it does not allow the flow of heat from the body. As a result, a new quilt is felt warmer than the old one.
l) A glass is filled with certain liquid at certain temperature such that it overflows whether heated or cooled.
A glass is filled with certain liquid at certain temperature such that it overflows either heated or cooled. The liquid is water at 4°C. It is because; water is a liquid which shows anomalous behavior in expansion. When water is heated from 0°C to 4°C, it contracts instead of expanding and when it is further heated beyond 4°C, it expands. Water has highest density at 4°C. Therefore, the given liquid is water at 4°C.
m) Two beakers A and B are full of water at 4°C. Beaker A is heated and B is cooled down, from which beaker water overflow? Why?
Water will overflow from both of the beakers because due to anomalous expansion of water, water has high density and least volume at 4°C, when it is heated it expands according to the normal expansion of water whereas when it is cooled it's volume increases due to anomalous expansion of water.
Heat Capacity
Heat capacity is defined as the amount of a heat energy required to raise the temperature of any mass of a substance through one degree Celsius or one Kelvin. Its SI unit is Joule per degree Celsius or Joule per Kelvin.
Specific Heat Capacity
Specific heat capacity is defined as the amount of heat energy required to raise the temperature of 1 kg of any substance through 1°C. Its SI unit is J/kg°C.
Meaning of Specific Heat Capacity of Water
Specific heat capacity of water is 4200 J/kg°C, its meaning is 4200 Joule heat energy is required to raise the temperature of 1 kg of water through 1°C.
Heat Equation
The heat lost or heat gained by any object is equal to the product of the mass of the object, specific heat capacity and change in temperature. This is called heat equation.
Mathematical Form
Q = m × S × Δt
Where,
Q = Amount of heat lost or heat gained by a body.
m = mass of the object.
S = specific heat capacity
Δt = change in temperature.
Calorimetry
Calorimetry is the branch of physics which deals with the measurement of quantities of heat.
Principle of Calorimetry
The principle of calorimetry states that the amount of heat lost by a hot body is equal to the heat gained by a cold body, if there is no loss of heat to the surroundings.
Heat lost by a hot body = Heat gained by a cold body
Proof of Q = mSΔt
According to the definition of the heat energy,
Heat energy gained or lost by a body (Q) is directly proportional to the mass (m) of the body
i.e. Q ∝ m ……(1)
Heat energy gained or lost by a body (Q) is directly proportional to the change in temperature (Δt) of the body,
i.e. Q ∝ Δt …….(2)
Combining eqn 1 and 2, we get,
Q ∝ mΔt
Q = S mΔt
Where, 'S' is a proportionality constant and is called as specific heat capacity of a substance.
Graph Analysis
[Graph showing volume and density changes of water with temperature would be placed here]
What does the first figure represent?
The first figure represents the variation of volume of water with temperature.
What does the second figure represent?
The second figure represents the variation of density of water with temperature.
What does this behavior of water called?
It represents anomalous expansion of water.
Write any two advantages of the above behavior.
Advantages:
- Fish and other aquatic animals can survive in the pond when water on the surface gets freezes.
- In cold countries, soft drink bottles can be stored below the frozen ice of the pond to prevent from freezing.
Give Reasons
The heat energy of two bodies having same temperature may be different.
The temperature of anybody depends upon the average K.E. of the molecules and is independent of the no. of molecules but heat energy depends upon the no. of molecules and K.E. of each molecule. So, according to the different mass the heat energy may be different in the bodies having same temperature.
A thick glass tumbler cracks when boiling water is kept in it.
When boiling water is kept in a glass tumbler, the lower part of the glass is heated so expands but since glass is insulator of heat, the upper part can't expands so it may leads to the cracking of glass.
What is the difference in the nature of the molecules of the water and ice?
Since the temperature of water is greater than ice the vibration of the molecules of the water is greater than the vibration of the molecules of the ice.
If 1kg of water and 1kg of mercury is heated with same amount of heat energy whose temperature is high? Why?
Mercury will have high temperature because its specific heat capacity is low.
Specific Heat Capacity Analysis
Questions:
- If equal amount of heat energy is supplied to equal mass of substances, which object will have high temperature and which have low temperature?
- If equal amount of heat energy is supplied to equal mass of substances which object have high rate of change of temperature?
- If the temperature of all three substance must be changed by 5°C to equal mass of substances, which one should be heated for longer time and why?
- If all the three substances of same mass and same temperature are left in the air which one will cool fast and why?
- If all the three substances of same temperature and same mass are placed above the slab of wax, which one will penetrate more and why?
- What is the meaning that specific heat capacity of X is 910 J/kg°C?
Answers:
- According to the heat equation Q = mSΔt, with Q and m constant, Δt is inversely proportional to S. Hence 'Y' will have high temperature and 'X' will have low temperature.
- Same as (i) - 'Y' will have the highest rate of temperature change.
- 'X' should be heated for longer time as it requires more heat energy to reach to 5°C due to its higher specific heat capacity.
- 'Y' will cool faster as it has the least specific heat capacity and heat energy.
- 'X' will penetrate more in the wax as it contains more heat energy due to its higher specific heat capacity.
- The specific heat capacity is 910 J/kg°C means 910 Joule heat energy is required to raise the temperature of 1 kg of X through 1°C.
Applications of Specific Heat Capacity
Water is used as a coolant in radiator of a vehicle.
The specific heat capacity of water is 4200 J/kg°C which is too much high. Water absorbs large amount of heat energy from the engine with less rise in temperature. So, water is used as a coolant in the radiator of the vehicle to cool engine.
Water is used in a hot water bag.
Hot water bag is used to heat the swollen part of the body for longer time. Since, specific heat capacity of water is high, the water remains hot for longer time. So, it is used in a hot water bag.
A wet handkerchief is kept on the forehead of the patient suffering from fever.
When a person is suffering from fever, a wet handkerchief is kept on forehead to lower his body temperature because the wet handkerchief contains water, whose specific heat capacity is high, absorbs large amount of heat energy with less rise in temperature of the water. So, it helps to reduce the temperature of the patients.
Sandy soil gets heated very quickly as compared to wet soil, why?
The specific heat capacity of water (4200 J/kg°C) is much more greater than the specific heat capacity of sand (800 J/kg°C). So, to the same rise of temperature water needs almost 5 times more heat energy than sand. So, sandy soil gets heated very quickly as compared to wet soil.
Well water is warmer in the morning than during winter season.
The specific heat capacity of soil (land surface) is much more less than that of the water. So, during winter night, the temperature of the soil decreases very fast than the water. As a result in the morning times heat from water starts to move from the well water. Hence, well water is felt warmer in the morning during winter season.
In desert, days are very hot and the night are very cold.
The surface of desert contains large amount of sand having specific heat capacity low(800 J/kg°C). So, due to less specific heat capacity sand becomes very hot during day by getting heat of the sun whereas the hot sand also looses it's heat very soon during night. So, in deserts, days are very hot and the nights are very cold.
The difference in temperature is low near the cities in the sea or islands.
Sea and ocean consists of a large amount of water having specific heat capacity high(4200 J/kg°C). Due to this it gets heated slowly during day and gets cooled very slowly during night. Therefore, the difference in temperature is very low near sea because of land breeze and sea breeze.
We feel more cold in a steel chair than in a wooden chair during winter.
The specific heat capacity of steel is less than wood. So, the steel chair looses heat energy fast and becomes cold faster than the wooden chair. So, we feel more cold in a steel chair than wooden chair during winter.
What is land breeze and sea breeze? Explain how these occur.
The blowing of air towards the land from the sea is called land breeze whereas the blowing of air towards the sea from land is called sea breeze.
The specific heat capacity of water is much greater than the sand. So, in the day time due to the sunlight, the temperature of the sand increases rapidly but the temperature of water near the sea increases slowly. So, the air starts to blow from sea to the land which is called sea breeze whereas during night, the temperature of water falls slowly and the temperature of sand falls rapidly causing the flow of air from land towards the sea, this is called land breeze.
Explain why the water kept in earthen pot remains cold.
The earthen pot contains different capillary pores. So, from these pores water comes out due to evaporation. The evaporating water takes heat from the water inside the pot. So, the water in the pot remains cooled.
Numerical Problems
1. Convert 40°C into °F.
Solution:
According to the relation of the temperature scale:
(C - 0)/100 = (F - 32)/180
Or, (40 - 0)/100 = (F - 32)/180
Or, 720 = 10F - 320
Or, 10F = 1040
Or, F = 104
Hence, 40°C = 104°F
2. Convert 2.356 Cal/g°C into J/Kg°C.
Solution:
We have, 1 cal/g°C = 4200 J/kg°C
Or, 2.356 cal/g°C = 4200 × 2.356 J/kg°C
= 9895.2 J/kg°C
Hence, 2.356 Cal/g°C = 9895.2 J/kg°C
3. The temperature of 20 Kg of water in the radiator of engine of a car is 30°C. If the temperature of water increases up to 100°C after the engine is heated, what is the quantity of heat absorbed by water?
Solution:
Mass of water (m) = 20kg
Specific heat capacity (S) = 4200 J/kg°C
Initial temperature (t₁) = 30°C
Final temperature (t₂) = 100°C
Difference in temperature (Δt) = t₂ - t₁ = 100 - 30 = 70°C
Heat energy (Q) = ?
We have, Q = mSΔt
Or, Q = 20 × 4200 × 70
Or, Q = 5.88 × 10⁶ J
Hence, 5.88 × 10⁶ Joule of heat energy is absorbed by the water.
4. 10 kg of water at the temperature of 50°C is mixed with 5 kg of water at the temperature of 80°C in a vessel. What will be the final temperature?
Solution:
For hot water:
Mass (m₁) = 5kg
Temperature (t₁) = 80°C
Specific heat capacity = s
For cold water:
Mass (m₂) = 10kg
Temperature (t₂) = 50°C
Specific heat capacity = s
Let the final temperature of mixture be 'T'
From principle of calorimetry:
Heat lost by hot water = Heat gained by cold water
Or, Q₁ = Q₂
or, m₁s(t₁ - T) = m₂s(T - t₂)
or, 5 × (80 - T) = 10 × (T - 50)
or, 80 - T = 2T - 100
or, 180 = 3T
or, T = 60°C
Hence, the final temperature of mixture is 60°C.
5. If a heater with a power 1000 W gives 3.6 × 10⁶ joules of heat energy in one hour. What will be the temperature raised in the 50 kg of water with temperature 30°C by the heat in one hour?
Solution:
Mass of water (m) = 50kg
Heat energy (Q) = 3.6 × 10⁶ J
Rise in temperature (Δt) = ?
Specific heat capacity (s) = 4200 J/kg°C
We have, Q = msΔt
Or, 3.6 × 10⁶ J = 50 × 4200 × Δt
Or, Δt = 3.6 × 10⁶ / 210000
Or, Δt = 17.14°C
Hence, Rise in temperature = 17.14°C
6. If a heater with a power 1000 W is operated for one hour to heat water. What will be the temperature raised in the 50 kg of water by the heat?
Solution:
Mass of water (m) = 50kg
Specific heat capacity (s) = 4200 J/kg°C
Power of heater (P) = 1000 W
Time taken (t) = 1hr = 60 × 60 seconds = 3600 seconds
We have, Power = Heat energy (Q) / time taken
Or, Heat energy (Q) = P × t
Or, Q = 1000 × 3600
Or, Q = 3.6 × 10⁶ J
Again from heat equation Q = msΔt
Or, 3.6 × 10⁶ = 50 × 4200 × Δt
Or, Δt = 17.14°C
Hence, Rise in temperature = 17.14°C
Define 1 joule heat energy
One joule heat energy is defined as the amount of the heat energy required to raise the temperature of any substance of mass 1 kg through 1°C having specific heat capacity 1 J/kg°C.







0 Comments